The checklist for new zoological genus and species names is published
February 25, 2026
A little over a month ago, I announced that DRAFT v4 of the the checklist for new zoological genus and species names, incorporating information about electronic publication, was available for critique. Perhaps surprisingly, there were no comments (in contrast to the 127 comments on DRAFT v3). So I have gone ahead and published the checklist. You are free to use this in your own work. If you want to cite it, I recommend:
- Taylor, Michael P., 2026. Checklist for new zoological genus and species names. Sauropod Vertebra Picture of the Week, 25 February, 2026. doi:10.59350/knt7m-whb54. https://svpow.com/2026/02/25/checklist-for-new-zoological-genus-and-species-names/ archived at https://web.archive.org/web/20260225210412/https://svpow.com/2026/02/25/checklist-for-new-zoological-genus-and-species-names/
(Note that this is DOI of this post, not of the checklist.)
If you believe in “Artificial Intelligence”, take five minutes to ask it about stuff you know well
February 14, 2025
Here’s a Mastodon thread from a year ago.
Just a quick check on how ChatGPT’s getting on …
Me: Who reassigned the species Brachiosaurus brancai to its own genus, and when?
ChatGPT: The species Brachiosaurus brancai was reassigned to its own genus, Giraffatitan, by the paleontologist Michael Janensch in 1914.
Me: Dude, that is so wrong I can’t even.
ChatGPT: I apologize for the mistake in my previous response. The reassignment of Brachiosaurus brancai to Giraffatitan brancai was not done by Michael Janensch. The correct attribution goes to the paleontologist Ernst Stromer, who reclassified it in 1914.
Me: That answer is even worse. Stromer had nothing to do with the genus name or species name.
ChatGPT: I apologize for the confusion in my previous responses. The reassignment of Brachiosaurus brancai to Giraffatitan brancai was actually done by the paleontologist Werner Janensch, not Michael Janensch, in 1914.
sigh
Regular readers will know that the actual sequence of events was:
- Riggs (1903) named the genus Brachiosaurus and its type species Brachiosaurus altithorax.
- Janensch (1914) named a second species of this genus, “Brachiosaurus” brancai.
- Paul (1988) considered the two species separate at the subgeneric level (a taxonomic level that is rarely if ever used in vertebrate palaeontology) and named the subgenus Brachiosaurus (Giraffatitan) brancai.
- Olshevsky (1991) used the genus-and-species combination Giraffatitan brancai without comment in a self-published booklet, a combinaton which was not followed by any formal publication known to me, until …
- Taylor (2009) demonstrated that numerous characters separate the two species and that Giraffatitan is a separate genus with type species G. brancai.
Now, ChatGPT got every single part of that wrong. It left Riggs, Paul, Olshevsky and me out completely. It credited Werner Janensch for reassigning a species to a genus whose name was coined 19 years after he died. It pulled poor Ernst Stromer in for some reason. It invented “the paleontologist Michael Janensch”, who is presumably the unholy offspring of Werner Janensch and me. And in all three answers, it insisted that the reassignment was done in 1914, 74 years before the name Giraffatitan was coined.
But the worst part of this is not the errors. It’s not the blithe confidence with which the false facts are recited. It’s not even the bland “I apologize for the mistake in my previous response” to be followed by more utter nonsense. It’s that these incorrect answers look so plausible. For a lay-person — someone who, foolishly, has not been reading this blog for the last eighteen years — the answers given here look superficially reasonable. A kid doing a homework report on Brachiosaurus could take these answers and weave them into the submission without even having an inkling that they’re completely wrong. And the teacher who has to mark the essay will also likely swallow them.
Because LLMs get catastrophically wrong answers on topics I know well, I do not trust them at all on topics I don’t already know. And if you do trust them, I urge you to spend five minutes asking your favourite one about something you know in detail.
Why do I keep banging on about this? Because we’ve reached an especially toxic phase in the history of AI. A lot of companies have ploughed billions of dollars into the dream of being able to replace human workers with machines, and they are desperate to make us believe it’s going to work — if only so they can cash out their investments while the stocks are still high. We’re in the “pump” phase of an industry-wide pump-and-dump. It would be wise for all to beware getting suckered in.
(Disclaimer: LLMs are useful for some classes of queries. I use them a lot to remind me of programming-language idioms, asking questions like “In Go, if I have an object satisfying the error interface, how can I determine which specific kind of error structure it is?”. They’re good for this because you can quickly determine whether the answer is correct or not, thanks to the merciless compiler. LLMs are not useless; they’re just way overhyped and misapplied.)
References
- Janensch, Werner. 1914. Ubersicht uber der Wirbeltierfauna der Tendaguru-Schichten nebst einer kurzen Charakterisierung der neu aufgefuhrten Arten von Sauropoden. Archiv fur Biontologie, Berlin, III, 1(1):81-110.
- Olshevsky, George. 1991. A Revision of the Parainfraclass Archosauria Cope, 1869, Excluding the Advanced Crocodylia. Mesozoic Meanderings #2 (1st printing): iv + 196 pp.
- Paul, Gregory S. 1988. The brachiosaur giants of the Morrison and Tendaguru with a description of a new subgenus, Giraffatitan, and a comparison of the world’s largest dinosaurs. Hunteria 2(3):1-14.
- Riggs, Elmer S. 1903. Brachiosaurus altithorax, the largest known dinosaur. American Journal of Science 15(4):299-306.
- Taylor, Michael P. 2009. A re-evaluation of Brachiosaurus altithorax Riggs 1903 (Dinosauria, Sauropoda) and its generic separation from Giraffatitan brancai (Janensch 1914). Journal of Vertebrate Paleontology 29(3):787-806.
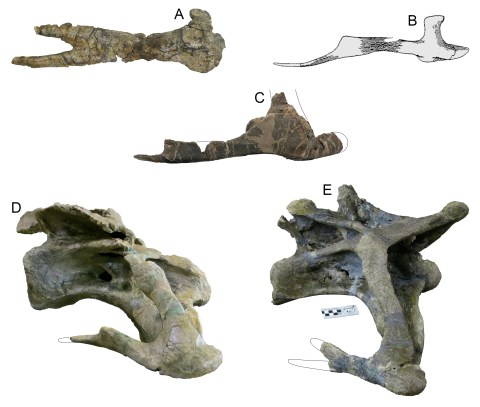
Here at SV-POW!, we love bifurcated cervical ribs. Those of Turiasaurus are one of the autapomorphies proposed by Royo-Torres et al. (2006:figure 1K). Their diagnosis of the new genus included “accessory process projecting caudodorsally from the dorsal margin of the shafts of proximal cervical ribs”. Here is the best example of such a rib in Turiasaurus, attached to its vertebra. (It’s a shame the black backdrop doesn’t extend beneath the rib, but you can make it out easily enough nevertheless.)

Cervical vertebra of the holotype individual of Turiasaurus riodevensis, in left lateral view. Photograph by Andrew Moore, reproduced with permission, and I am delighted to get this out there!
This is a very similar but not identical photo to that used in Royo-Torres et al.’s (2006:figure 1K) illustration; but because that paper was unfortunately published in Science instead of in a scientific journal, the illustration is microscopic and the description perfunctory. There are no further illustrations of the material in the supplementary document.
Aaanyway. We mentioned but did not illustrate this rib in our recent paper (Wedel and Taylor 2023), and we wrote of it (on page 93) that:
Royo-Torres et al. (2006) described and illustrated bifurcated cervical ribs in Turiasaurus, and Britt et al. (2017) described and illustrated bifurcated cervical ribs in Moabosaurus (Fig. 3A). Turiasaurus and Moabosaurus are both members of the clade Turiasauria, but bifurcated ribs are absent in Mierasaurus, which Royo-Torres et al. (2017) recovered as the sister taxon of Moabosaurus within Turiasauria. This implies either a single origin of bifurcated cervical ribs in Turiasauria, with a reversal in Mierasaurus, or parallel origins of bifurcated cervical ribs in Turiasaurus and Moabosaurus. [emphasis added]
I think we missed the most obvious explanation here: that while the potential to develop bifurcated cervical ribs is phylogenetically determined, the actual development of bifurcation in any given rib is highly variable between individuals and indeed between vertebrae of a single individual. Given that we showed this individual variability pretty clearly for apatosaurines — within the Apatosaurus louisae holotype CM 3018, for example — it’s a bit dumb that we failed to apply the same observation to the variable appearance of bifurcated cervical ribs in turiasaurs.
Anyway, that’s not why we’re here. We’re here to look again at how different the bifurcation is between Turiasaurus and some apatosaurines. Here’s a composite, based on the photo above and our old friend MWC 1946 (also appearing as Wedel and Taylor 2023:figure 3D).

Top: Turiasaurus riodevensis, left rib of unspecified cervical vertebra with unknown specimen number, left lateral view. Bottom: Apatosaurinae indet. MWC 1946, right cervical rib in right lateral view, reversed.
(Why don’t we know the specimen number of the Turiasaurus cervical? Again, because the “description” of this important and unusually complete sauropod was published in Science, which is far too important to waste space on trivia like specimen numbers. All we’re told is that the holotype elements have specimen numbers in the range CPT-1195 to CPT-1210. Once more, Science is the opposite of science. Digression ends.)
Back in November last year, Matt asked the question: Single-segment neck muscles in diplodocids?. He tentatively concluded yes, based on the posterodorsal trajectory of the upper prong in apatosaurine cervical ribs, which would have anchored short flexor colli lateralis muscles attaching to the cervical rib loop of the immediate successor vertebra. (We know where on the vertebra these muscles originate in birds, and the upward-and-backward orientation of the prong points to where that site is on the very next vertebra.)
Looking at the composite image above suggests that the same was not true in Turiasaurus. Here, the upper and lower prongs of the bifurcated rib are close to parallel, implying that flexor colli lateralis muscles inserting on the upper prongs can only have originated some segments further back.
Now if we return to the Freak Gallery in our recent paper:
Wedel and Taylor 2023:Figure 3. Bifurcated and incipiently bifurcated cervical ribs of sauropods. A, Moabosaurus utahensis holotype individual, left cervical rib BYU 14063 (not right as stated by Britt et al. 2017), probably associated with C5, in medial view. B, Dicraeosaurus hansemanni holotype MB.R.2379, right cervical rib 8 in lateral view. Modified from Janensch (1929, fig. 21). C, Brontosaurus parvus CM 555, right cervical rib 7 in lateral view. D, Apatosaurus louisae MWC 1946, cervical vertebra in right lateral view. E, Apatosaurus louisae MWC 5659, cervical vertebra in left lateral view (reversed). All photographs by the authors.
We can see that the rib of Moabosaurus has near-parallel prongs like those of its relative Turiasaurus — implying that it, too, likely had multi-segment flexor colli lateralis muscles. But by contrast the Dicraeosaurus rib, which we described as “incipiently bifurcated”, has a more or less dorsally projecting flange which seems likely to have anchored a single-segment muscle as in apatosaurines.
So my tentative conclusion is that bifurcated ribs in diplodocids (such as apatosaurines and Dicraeosaurus) do indeed imply single-segment neck muscles (or at least single-segment flexor colli lateralis); whereas the bifurcated ribs of turiasaurians imply multi-segment muscles.
Admittedly this is a biggish conclusion to hang on such scant evidence as the apparent angle of divergence in the handful of bifurcated cervical ribs that we’ve seen. But I think it at least stands as the hypothesis best supported by presently available evidence, and it’s there to be corroborated or contradicted by further observations.
References
- Royo-Torres, Rafael, Alberto Cobos and Luis Alcalá. 2006. A giant European dinosaur and a new sauropod clade. Science 314:1925–1927.
- Wedel, Mathew J., and Michael P. Taylor. 2023. The biomechanical significance of bifurcated cervical ribs in apatosaurine sauropods. VAMP (Vertebrate Anatomy Morphology Palaeontology) 11:91-100. doi: 10.18435/vamp29394
3D printing is especially useful for sauropod workers
September 28, 2022
This is the first 3D print of a dinosaur bone that I ever had access to: the third caudal vertebra of MWC 8028, the ‘new’ Haplocanthosaurus specimen from Snowmass, Colorado (Foster and Wedel 2014, Wedel et al. 2021). I’ve been carrying this thing around since 2018. It’s been an aid to thought. I touched on this before, in this post, but real sauropod vertebrae are almost always a giant pain to work with, given their charming combination of great weight, fragility, and irreplaceability. As opposed to scaled 3D prints, which are light, tough, and endlessly replaceable.
This was brought home to me again a couple of weeks ago, when I visited the Carnegie Museum, in Pittsburgh, Pennsylvania, and Research Casting International, in Trenton, Ontario, Canada. I was at each place to have another look at their haplocanthosaur specimens. The Carnegie is of course the home of CM 572, the type of H. priscus, and CM 879, the type of H. utterbacki (which has long been sunk into H. priscus, and rightly so — more on that another time, perhaps). RCI currently has CMNH 10380, the holotype of H. delfsi, for reprepping and remounting before it goes back to the Cleveland Museum of Natural History.
The caudals of CM 572 and CM 879 aren’t that different in size — the centra max out at about 20cm (8in) in diameter, and the biggest, caudal 1 of CM 572, is 50cm (20in) tall. Still, given their weight and the number of thin projecting processes that could possibly break off, I handled them gingerly.
The caudals of H. delfsi are a whole other kettle of fish. Caudal 1 has a max diameter of 36cm (14in) and a total height of 85cm (33.5in). I didn’t handle that one by myself unless I absolutely had to. Fortunately Garth Dallman of RCI helped a lot with the very literal heavy lifting, as did fellow researcher Brian Curtice, who was there at the same time I was.
Back to my beloved MWC 8028, the Snowmass haplocanthosaur. My colleagues and I are still working on it, and there will be more papers coming down the pike in due time (f’rinstance). I’m pretty sure that the main reason we’ve been able to get so much mileage out of this mostly incomplete and somewhat roadkilled specimen is that we’ve had 3D prints of key bones to play with. Now, I joke all the time about being a grownup who gets paid to play with dinosaur bones, but for once I’m not writing in jest when I say ‘play with’. That 3D printed caudal is basically a dinosaurian fidget toy for me, and I think it’s probably impossible to play with anatomical specimens without getting interested in their nooks and crannies and bits and bobs.
Another nice thing about it: I can throw it in my luggage, take it Oklahoma or Utah or Pennsylvania or Canada, and just plop it in someone’s hand and say, “Look at this weird thing. Have you ever seen that before?” I have done that, in all of those places, and it’s even more convenient and useful than showing CT slices on my laptop. I’ve watched my friends and colleagues run their fingers over the print, pinch its nearly non-existent centrum, poke at its weird neural canal, and really grokk its unusual morphology. And then we’ve had more productive conversations than we would have otherwise — they really Get It, because they’ve really handled it.
When I started writing this post, the title was a question, but that’s tentative to the point of being misleading. Three-D prints are obviously useful for sauropod workers because with very few exceptions our specimens are otherwise un-play-with-able. And playing with dinosaur bones turns out to be a pretty great way to make discoveries, and to share them.
(And yes, we’ll be publishing the CT scans and 3D models of MWC 8028 in due time, so you can play with it yourself.)
References
- Foster, J.R., and Wedel, M.J. 2014. Haplocanthosaurus (Saurischia: Sauropoda) from the lower Morrison Formation (Upper Jurassic) near Snowmass, Colorado. Volumina Jurassica 12(2): 197–210. DOI: 10.5604/17313708 .1130144
- Wedel, Mathew; Atterholt, Jessie; Dooley, Jr., Alton C.; Farooq, Saad; Macalino, Jeff; Nalley, Thierra K.; Wisser, Gary; and Yasmer, John. 2021. Expanded neural canals in the caudal vertebrae of a specimen of Haplocanthosaurus. Academia Letters, Article 911, 10pp. DOI: 10.20935/AL911

Morphological variation in paramedullary airways; yellow = spinal cord, green = diverticula. The spectrum of variation is discretized into four groups: i, branches of intertransverse diverticula contact spinal cord at intervertebral joints; ii, branches of intertransverse diverticula extend partially into the vertebral canal, but remain discontinuous; iii, paramedullary diverticula form sets of tubes that are continuous through vertebral canals of at least two consecutive vertebrae; iv, continuous paramedullary diverticula anastomose with supravertebral diverticula. Each variant is depicted diagrammatically (A, dorsal view; B, E, H, & K, transverse view) and shown in two CT scans; images in each column correspond to the same morphology. Morphology i: C, cormorant; D, scrub jay. Morphology ii: F, bushtit; G, common murre. Morphology iii: I, red-tailed hawk; J, black-crowned night heron. Morphology iv: L, M, pelican. (Atterholt and Wedel 2022: figure 5)
New paper out:
Quick aside, which will soon be of historical interest only: so far, only the accepted-but-unformatted manuscript is available, with the final, fully-formatted ‘version of record’ due along at some point in the future. We’re not sure when that will be — could be next week, could be months from now — which is why I’m following my standard procedure and yapping about the new paper now. This has paid off in the past, when papers that were only available in accepted ms form were read and cited before the final version was published. UPDATE on April 9: the formatted version of record is out now, as an open-access article with a CC-BY license, and I swapped it for the ‘accepted ms’ version in the links above and at the end of this post.
This paper has had a weirdly drawn-out gestation. Jessie and I hatched the idea of it way back in 2017, when we were teaching in the summer anatomy course together. I learned that Jessie had a big war chest of CTs of dead birds, and I’d been obsessed with supramedullary diverticula in birds and sauropods for some time already (e.g., an SVPCA talk: Wedel et al. 2014). There were detailed published descriptions of the supramedullary diverticula in a handful of species — namely chickens, turkeys, and pigeons — but no broad survey of those diverticula across living birds. Jessie had the CT scans to do that big survey, which we got rolling on right away. She presented our preliminary results at SVPCA in 2018 (Atterholt and Wedel 2018), and by rights the paper should have been along shortly thereafter. More on that in a sec.
One thing that may seem odd: we use the term ‘paramedullary diverticula’ instead of the more familiar and established ‘supramedullary diverticula’. That’s because these diverticula are not always dorsal to the spinal cord; sometimes they’re lateral, sometimes they’re ventral, and sometimes they completely surround the spinal cord, like an inflated cuff. So we decided that the term ‘paramedullary’, or ‘next to the spinal cord’, was more accurate than ‘supramedullary’, or ‘above the spinal cord’, for describing this class of diverticula.

Observed variation in the shape, arrangement, and orientation of paramedullary diverticula relative to the spinal cord; yellow = spinal cord, green = diverticula. A, paired diverticula dorsal to spinal cord in an ostrich. B, paired diverticula lateral to spinal cord in a bushtit. C, paired diverticula ventral to spinal cord in a violet turaco. D, three diverticula dorsal to spinal cord in an ostrich. E, four diverticula dorsal to spinal cord in an eclectus parrot. F, single, c-shaped diverticulum dorsal to spinal cord in an ostrich. G, diverticula completely surrounding spinal cord and pneumatizing vertebra in a violet turaco. H, no paramedullary diverticula present in a Pacific loon. I, diverticula completely surrounding spinal cord in a pelican. (Atterholt and Wedel 2022: figure 6)
I will have more to say about the science in other posts, and you can get the scientific backstory in this post and this one and the abstracts cited above and linked below. The rest of this post is mostly about me, so if you stick around, buckle up for some advanced navel-gazing.
There’s no one reason why this paper didn’t come out sooner. In short, I hit a wall. We went through a curriculum change at work, and suddenly the annual schedule that I’d relied on for a decade was completely upended. I had some unexpected challenges in my personal life. But the biggest problem was that my attitude toward research and writing had changed, for the worse.
When I was fresh out of grad school I had this kinda snotty attitude that my research was MINE, and wherever I was teaching was just, like, a paycheck, man, but they don’t own me, or my research. And as my teaching and committee responsibilities ramped up I still felt like research and writing was something I did for myself, and that my mission was to steal however many hours I could away from the “day-job work” to get done the things that I really wanted to do. Like a guerilla insurgency. In retrospect, it was a pretty good attitude for getting stuff done.
But somewhere along the way, I stopped thinking about research as something that belonged to me, something that I did for myself, and started thinking about it as part of my job. (This also maybe is not so flattering in what it reveals about how I think, or at least thought, about my job.) Instead of using my research time as a source of energy and a wellspring of satisfaction and positivity, I starting thinking of it only as a sink. And it happened so insidiously that I didn’t even realize it. My productivity plummeted, and I didn’t understand why. I was restless and depressed, and I didn’t understand that either. At the level of my superficial thoughts I still wanted to get research done, but my subconscious was turned off to it, so I just spun my wheels.
Then the pandemic hit. I’d always been a pretty optimistic, upbeat person, but I found myself just staring off into space franticizing about all the horrible things going on in the world, or staying up too late doom-scrolling the news. I slept too little, and poorly, and by the end of 2020 I felt worn down to a nub.

Osteological evidence of paramedullary diverticula. A, pocked texturing inside the vertebral canal of a pelican (LACM 86262). B, pneumatic foramen on the roof of the vertebral canal of an albatross (Phoebastria nigripes, LACM 115139). C, pneumatic foramina in the floor of the vertebral canal of an ostrich (Struthio camelus, LACM 116205). D, deep pneumatic fossae in the roof of the vertebral canal of an Eastern moa (Emeus sp., LACM unnumbered). (Atterholt and Wedel 2022: figure 7)
Then a series of positive things happened:
- I received a long, heartfelt email from Jessie (fittingly!), asking after me and laying out a plan for getting the paper done and out. It was the kick I needed to look inside and start picking myself apart, to figure out what the heck was going on. Much of this post is cribbed from my reply to her.
- I got a little break from lecturing in the spring of 2021, and that gave me the space to get a couple of things finished and submitted — the pneumatic variation paper with Mike in January (Taylor and Wedel 2021), and the Haplocanthosaurus neural canal paper, which was submitted even earlier in January, although it came out much later (Wedel et al. 2021; more on that publication delay in a future post).
- Finally, I had young, energetic coauthors who were moving projects forward that required modest levels of effort from me, but which paid off with highly visible publications that I’m proud to be an author on, including the saltasaur pneumaticity paper (Aureliano et al. 2021) and the ‘Sauro-Throat’ paper (Woodruff et al. 2022).
It’s impossible to overstate how energizing it was to get new things done and out, and how much it helped to have collaborators who were putting wins on the board even when I was otherwise occupied. One of those collaborators was Jessie, who just kept pushing this thing forward — and, sometimes, pushing me forward — until it was done. So the paper you can read today is a testament not only to her acumen as a morphologist, but also to her tenacity as a scholar, and as a friend.
The part of the paper I’m happiest about is the “Conclusions and Directions for Future Research”, which points the way toward a LOT of further studies that need to be done, both on extant birds and on fossil archosaurs, ranging from bone histology to functional morphology to macroevolution. As we wrote in the concluding sentence of the paper, “We hope that this study serves as a foundation and an enticement for further studies of this most unusual anatomical system, in both extinct and extant archosaurs.”
I can’t wait to see what comes next.
References
- Atterholt, J., and Wedel, M. 2018. A CT-based survey of supramedullary diverticula in extant birds. 66th Symposium on Vertebrate Palaeontology and Comparative Anatomy, Programme and Abstracts, p. 30.
- Atterholt, Jessie, and Wedel, Mathew J. 2022. A computed tomography-based survey of paramedullary diverticula in extant Aves. The Anatomical Record 306(1): 29-50. https://doi.org/10.1002/ar.24923
- Aureliano, Tito, Aline M. Ghilardi, Bruno A. Navarro, Marcelo A. Fernandes, Fresia Ricardi-Branco, & Mathew J. Wedel. 2021. Exquisite air sac histological traces in a hyperpneumatized nanoid sauropod dinosaur from South America. Scientific Reports 11: 24207.
- Taylor, Michael P., and Mathew J. Wedel. 2021. Why is vertebral pneumaticity in sauropod dinosaurs so variable? Qeios 1G6J3Q. doi:10.32388/1G6J3Q.5
- Wedel, M.J., Fiorillo, A., Maxwell, D., and Tykoski, R. 2014. Pneumatic diverticula associated with the spinal cord in birds, sauropod dinosaurs, and other ornithodiran archosaurs. 62nd Symposium on Vertebrate Palaeontology and Comparative Anatomy, Meeting Proceedings, p. 60.
- Wedel, Mathew; Atterholt, Jessie; Dooley, Jr., Alton C.; Farooq, Saad; Macalino, Jeff; Nalley, Thierra K.; Wisser, Gary; and Yasmer, John. 2021. Expanded neural canals in the caudal vertebrae of a specimen of Haplocanthosaurus. Academia Letters, Article 911, 10pp. DOI: 10.20935/AL911
- Woodruff, D. Cary, Wolff, Ewan D.S., Wedel, Mathew J., Dennison, Sophie, and Witmer, Lawrence M. 2022. The first occurrence of an avian-style respiratory infection in a non-avian dinosaur. Scientific Reports 12, 1954. https://doi.org/10.1038/s41598-022-05761-3
Early origin of artiodactyls in South America
February 9, 2021
Bonaparte’s (1999) description of the Early Cretaceous sauropod Agustinia ligabuie was notable for its identification of nine bony fragments as representing dermal armour, which he classified into Types 1–4. Here are some examples:

Bonaparte 1999: figure 3. Agustinia ligabuei gen et sp. nov. Osteoderms. A, Type 1. B, Type 2. C, Type 3. Abbrev.: po, thick proximal ossification.
Consequently, Augustinia was for many years restored as uniquely spiky, a sort of “stegosaur sauropod”:
But Bellardini and Cerda (2017) showed that the so-called “osteoderms” are probably no such thing, but represent other, more normal, bony elements. Bonaparte’s types 1, 3 and 4 were all reinterpreted as complete or partial dorsal ribs, and type 2 as a portion of the iliac blade:

Bellardini and Cerda 2017: figure 6. Bony element type 2 (MCF-PVPH-110/08) of A. ligabuei, in medial (A1–2), and lateral views (B1–2) with interpretive line drawings. In this work we propose that MCF-PVPH-110/08 is a dorsal portion of the left iliac blade, centered above the acetabulum (C). Abbreviations: dmil dorsal margin of iliac blade, poap post-acetabular process, prap pre-acetabular process, sr sacral rib. Dashed lines indicate missing bone. Asterisks indicate the hypothetical position of morphological features.
However, careful re-examination of Bellardini and Cerda 2017: figure 6: part A1 shows clearly that the so-called type-2 osteoderm is in fact a sheep:
This is a significant finding, as the origin of Artiodactyla is generally held to have occurred in the early Eocene, with the earliest known representatives being from Europe, Asia, and North America (Rose 1996). The reidentification of the A. ligabuei type-2 osteoderm as a sheep pushes back the origin of artiodactyls by some 60 million years to the Aptian or Albian, and locates South America as the continent of origin.
References
- Bellardini, Flavio, and Ignacio A. Cerda. 2017. Bone histology sheds light on the nature of the “dermal armor” of the enigmatic sauropod dinosaur Agustinia ligabuei Bonaparte, 1999. The Science of Nature 104(1):1-13. doi:10.1007/s00114-016-1423-7
- Bonaparte, Jose F. 1999. An armoured sauropod from the Aptian of northern Patagonia, Argentina. pp. 1-12 in Y. Tomida, T. H. Rich and P. Vickers-Rich (eds.), Proceedings of the Second Gondwanan Dinosaur Symposium, Tokyo National Science Museum Monograph 15. Tokyo National Science Museum, Tokyo. x+296 pp.
- Rose, Kenneth D. 1996. On the origin of Artiodactyla. Proceedings of the National Academy of Sciences 93(4):1705-9. doi:10.1073/pnas.93.4.1705
The Snowmass Haplocanthosaurus project is now a museum exhibit
November 16, 2019

A life-size silhouette of the Snowmass Haplocanthosaurus, with Thierra Nalley, me, and Jessie Atterholt for scale. Photo by Jeremiah Scott.
Tiny Titan, a temporary exhibit about the Snowmass Haplocanthosaurus project, opened at the Western Science Center in Hemet, California, last night. How? Why? Read on.
Things have been quieter this year on the Haplo front than they were in 2018, for many reasons. My attention was pulled away by a lot of teaching and other day-job work–we’re launching a new curriculum at the med school, and that’s eaten an immense amount of time–and by some very exciting news from the field that I can’t tell you about quite yet (but watch this space). Things are still moving, and there will be a paper redescribing MWC 8028 and all the weird and cool things we’ve learned about it, but there are a few more timely things ahead of it in the queue.
One of the things going on behind the scenes this year is that Jessie Atterholt, Thierra Nalley, and I have been working with Alton Dooley, the director of the Western Science Center, on this exhibit. Alton has had a gleam in his eye for a long time of using the WSC’s temporary exhibit space to promote the work of local scientists, and we had the honor of being his first example. He also was not fazed by the fact that the project isn’t done–he wants to show the public the process of science in all of its serendipitous and non-linear glory, and not just the polished results that get communicated at the end.
Which is not to say that the exhibit isn’t polished. On the contrary, it looks phenomenal. Thanks to a loan from Julia McHugh at Dinosaur Journey in Colorado, most of the real fossils are on display. We’re only missing the ribs and most of the sacrum, which is too fragmentary and fragile to come out of its jacket. As you can see from the photo up top, there is a life-size vinyl silhouette of the Snowmass Haplo, with 3D prints of the vertebrae in approximate life position. Other 3D prints show the vertebrae before and after the process of sculpting, rescanning, and retrodeformation, as described in our presentation for the 1st Palaeontological Virtual Congress last year (that slideshow is a PeerJ Preprint, here). The exhibit also includes panels on “What is Haplocanthosaurus” and its relationships, on pneumaticity in sauropods, on the process of CT scanning and 3D modeling, and on the unusual anatomical features of the Snowmass specimen. The awesome display shown above, with the possibly-bumpy spinal cord and giant intervertebral discs reconstructed, was all Alton–he did the modeling, printing, and assembly himself.

Haplo vs Bronto. Thierra usually works on the evolution and development of the vertebral column in primates, so I had to show her how awesome vertebrae can be when they’re done right. Photo by Brittney Stoneburg.
My favorite thing in the exhibit is this striking comparison of one the Snowmass Haplo caudals with a proximal caudal from the big Oklahoma apatosaurine. This was Alton’s idea. He asked me if I had any photos of caudal vertebrae from really big sauropods that we could print at life size to compare to MWC 8028, and my mind went immediately to OMNH 1331, which is probably the second-largest-diameter vertebra of anything from all of North America (see the list here). It was also Alton’s idea to fill in the missing bits using one of Marsh’s plates of Brontosaurus excelsus from Como Bluff in Wyoming. It’s a pretty amazing display, and it turns out to have been a vehicle for discovery, too–I didn’t realize until I saw the verts side-by-side that the neural canal of the Snowmass Haplo caudal is almost as big as the neural canal from the giant apatosaurine caudal. It’s not a perfect comparison, because the OMNH fossil doesn’t preserve the neural canal, and the Como specimen isn’t that big, but proportionally, the Snowmass Haplo seems to have big honkin’ neural canals, not just at the midpoint (which we already knew), but all the way through. Looks like I have some measuring and comparing to do.
(Oh, about the title: we don’t know if the Snowmass Haplo was fully grown or not, but not all haplocanthosaurs were small. The mounted H. delfsi in Cleveland is huge, getting into Apatosaurus and Diplodocus territory. Everything we can assess in the Snowmass Haplo is fused, for what that’s worth. We have some rib chunks and Jessie will be doing histo on them to see if we can get ontogenetic information. I’ll keep you posted.)

Brian’s new Haplocanthosaurus restoration, along with some stinkin’ mammals. Photo by Jessie Atterholt.
Brian Engh contributed a fantastic life restoration of Haplocanthosaurus pro bono, thanks to this conversation, which took place in John Foster’s and ReBecca Hunt-Foster’s dining room about a month ago:
Brian: What are you drawing?
Me: Haplocanthosaurus.
Brian: Is that for the exhibit?
Me: Yup.
Brian (intense): Dude, I will draw you a Haplocanthosaurus.
Me: I know, but you’re a pro, and pros get paid, and we didn’t include a budget for paleoart.
Brian (fired up): I’m offended that you didn’t just ask me to draw you a Haplocanthosaurus.
Then he went to the Fosters’ couch, sat down with his sketchbook, and drew a Haplocanthosaurus. Not only is it a stunning piece on display in the exhibit, there are black-and-white printouts for kids to take and color (or for adults to take to their favorite tattoo artists, just sayin’). [Obligatory: this is not how things are supposed to work. We should all support original paleoart by supporting the artists who create it. But Brian just makes so damn many monsters that occasionally he has to kick one out for the heck of it. Also, I support him on Patreon, and you can, too, so at a stretch you could consider this the mother of all backer rewards.]
One special perk from the opening last night: Jessica Bramson was able to attend. Who’s that, you ask? Jessica’s son, Mike Gordon, found the first piece of bone from the Snowmass Haplo on their property in Colorado over a decade ago. Jessica and her family spent two years uncovering the fossils and trying to get paleontologists interested. In time she got in touch with John Foster, and the rest is history. Jessica lives in California now, and thanks to John’s efforts we were able to invite her to the exhibit opening to see her dinosaur meet the world. Stupidly, I did not get any photos with her, but I did thank her profusely.

A restored, retrodeformed caudal of the Snowmass Haplocanthosaurus, 3D-printed at life size for the exhibit. Photo swiped from the WSC Facebook page.
I owe a huge thanks to Alton Dooley for taking an interest in our work, and to the whole WSC crew for their hard work creating and promoting the exhibit. You all rock.
The exhibit will run through the end of March, 2020, at least. I deliberately did not show most of it, partly because I was too busy having fun last night to be diligent about taking photos, but mostly because I want you to go see it for yourself (I will do a retrospective post with more info after the exhibit comes down, for people who weren’t able to see it in person). If you make it out to Hemet, I hope you have half as much fun going through the exhibit as we did making it.
It pays to stay humble before nature
April 17, 2018
At the 2011 sauropod gigantism symposium in Bonn, John Hutchinson gave a talk on biomechanics of large animals. At the end he showed a short video of a rhino running full-tilt, tripping, and literally flipping end over end. After the wipeout, the rhino got up and trotted off, apparently unhurt. I don’t remember John’s exact words, but they were something like, “We have no idea how this happens – all our models suggest that rhino should be a pile of broken bones.”
His point was, there is a lot we don’t yet understand, and it pays to keep that in mind.
This morning Mike and I were discussing the Middlesborough meteorite, which fell to ground in front of some railway workers in 1881 and was reported be warm to the touch right after falling. Here’s a short, lightly-edited recap of our conversation.
Mike: If it had slowed to such a very mundane speed, due to atmospheric braking, how on earth was it only “new-milk warm” to the touch?
Me: You mean, since it was obviously melted to hell on its way in, how did it cool down so much? From what I’ve been able to find out so far, most small meteors that survive to hit the ground have finished aerobraking when they’re still about 50,000 feet up, and from there they experience a “dark fall” where they’re just a chunk of rock falling at terminal velocity. It’s pretty cold at 25,000 feet, much less 50,000 feet, so I assume a lot of the heat is pulled away by convection into cold air during the dark fall period. There are meteorites that have fallen onto lake or sea ice and not produced any apparent melting, so at least some of them do fall cold.
Mike: This makes sense. So we imagine that is was a lot hotter than that a short time before the impact, but that it cooled as it continued to fall.
This is an interesting example of how thoroughly physical intuition can mislead when you’re dealing with objects operating in unfamiliar realms: very big (e.g. the K/Pg impactor “splashing”), very small (e.g. insects “swimming” through air) or very fast (e.g. this meteorite’s temperature). Physics works over a huge range, and we only have developed intuition for a small sub-range.
Me: Your point about physics is well taken. The older I get, the more humility I have before nature, after seeing many, many things that I would have thought impossible.
The specific example I had in mind there was digging dinosaurs – if you’d asked me back when if any dinosaurs made burrows, I would have bet heavily against it. And yet, there they are.
Stay humble, folks. It’s weird out there.
Back in 2009, I posted on a big cervical series discovered in Big Bend National Park. Then in 2013 I posted again about how I was going to the Perot Museum in Dallas to see that cervical series, which by then was fully prepped and on display but awaiting a full description. Ron Tykoski and Tony Fiorillo (2016) published that description a couple of years ago, and after almost five years it’s probably time I posted an update.
I did visit the Perot Museum in 2013 and Ron and Tony kindly let me hop the fence and get up close and personal with their baby. I got a lot of nice photos and measurements of the big specimen. It’s an impressive thing. Compared to the other big sauropod cervicals I’ve gotten to play with, these vertebrae aren’t all that long – the two longest centra are about 80cm, compared to ~120cm for Sauroposeidon, Puertasaurus, and Patagotitan, and 137cm for Supersaurus (more details here) – but they are massive. According to the table of measurements (yay!) in Tykoski and Fiorillo (2016), which accord well with the measurements I took when I was there, the last vert is 117.5cm tall from the bottom of the cervical rib to the top of the neural spine, 98.4cm wide across the diapophyses, and has a cotyle measuring 29cm tall by 42cm wide. Here it is with me for scale:
I guarantee you, standing next to that thing and imagining it being inside the neck of a living animal is a breathtaking experience.
I failed in my mission in one way. In a comment on my 2013 post, I said, “I’ll try to get some good lateral views of the mount with as little perspective as possible.” But it can’t be done – the geometry of the room and the size of the skeleton don’t allow it, as Ron noted in the very next comment. There is one place in the exhibit hall where you can get the whole skeleton into the frame, and that’s a sort of right anterolateral oblique view. Here’s my best attempt:
So, this is an awesome specimen and you should go see it. As you can see from the photos, the vertebrae are right on the other side of the signage, with no glass between you and them, so you can see a lot. The rest of the exhibits are top notch as well. Definitely worth a visit if you find yourself within striking distance of Dallas.
Reference
Tykoski, R.S. and Fiorillo, A.R. 2016. An articulated cervical series of Alamosaurus sanjuanensis Gilmore, 1922 (Dinosauria, Sauropoda) from Texas: new perspective on the relationships of North America’s last giant sauropod. Journal of Systematic Palaeontology 15(5):339-364.
Posterior cervical vertebra of a juvenile Diplodocus
September 11, 2017
We’ve not done many picture-of-the-week posts here recently. Let’s change that! Here’s a lovely little specimen that we saw in BYU on the 2016 Sauropocalypse trip.

Wedel and Taylor (2013), Figure 7. BYU 12613, a posterior cervical of Diplodocus, in dorsal (top), left lateral (left), and posterior (right) views. It compares most favourably with C14 of D. carnegii CM 84/94 (Hatcher, 1901: plate 3) despite being only 42% as large, with a centrum length of 270 mm compared to 642 mm for C14 of D. carnegii.
(At least, this is catalogued as Diplodocus. Jaime Headden suggested, and Emanuel Tschopp corroborated, the idea that it’s more likely Kaatedocus.)












